Light-induced assembly and repeatable actuation in Ca2+-driven chemomechanical protein networks
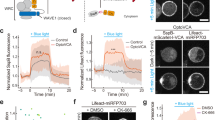
Researchers engineered a light-controlled chemomechanical network based on Tetrahymena Tcb2, a Ca2+-sensitive contractile protein, achieving rapid, repeatable self-assembly and contraction at rates comparable to actomyosin. Light-sensitive chelators permit precise optical Ca2+ release for spatiotemporal control, enabling active particle transport and seconds-scale actuation programmed with reinforcement learning; a coupled reaction-diffusion–elastic model explains observed dynamics. The platform is proposed for applications in synthetic cells, subcellular force generation, and programmable biomaterials.
Why it mattersThe Tcb2 Ca2+-responsive protein enables optically controlled contractility, informing biomaterials product design timelines.
